Automating the squeeze: Tolomatic prototype improves on hand-held emergency ventilator for COVID-19 patients
By Andrew Zaske on April 28, 2020
At Tolomatic, we are paying close attention to the need for medical device equipment to help with patient care during the COVID-19 crisis. There are hundreds of open-source designs and ideas on the Internet; we have reviewed many and considered how linear motion might improve on them. One design in particular caught our attention because it can be easily automated—with benefits for both the patient and operator. Our prototype automates a hand-held, portable and non-invasive emergency ventilator. A hand-held device designed for an emergency application has some drawbacks for extended use, but adding linear motion gives what we think is a reliable, safe and cost-effective improvement. Hence, “automating the squeeze.”
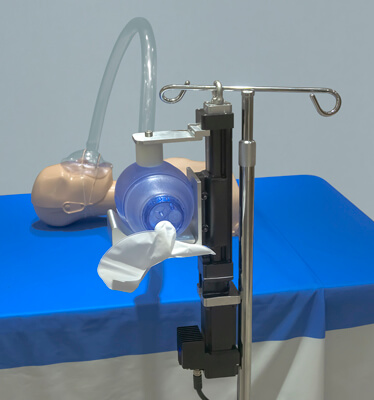
The emergency non-invasive, positive pressure ventilator is different from the expensive and invasive internal ventilators that we have become used to seeing in hospital settings. These internal ventilators require a patient to be intubated and the tube inserted down the throat. A portable, non-invasive emergency ventilator delivers air into the lungs of patients who are not breathing or whose breathing requires assistance. Unlike a tube down the throat for the invasive ventilator, the non-invasive method is to place a mask over the patient’s nose and/or mouth and use periodic forced air “puffs” to inflate the lungs.
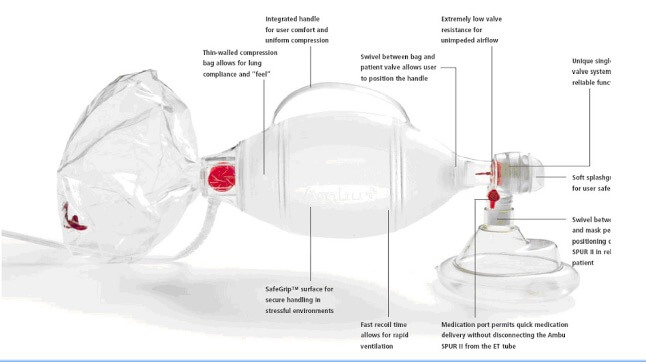
There are many names for this type of non-invasive emergency ventilator. Its trademark name is ambu bag, for artificial manual breathing unit (and that term is now used like Kleenex is for tissues). The generic name is bag-valve mask (BVM) or self-inflating bag. Medical professionals sometimes use the term “bagging the patient” to describe the procedure.
It’s a simple device, consisting primarily of the oxygen bag, tubes, mask and an oxygen inlet to supplement ambient air. Typically, an assistant squeezes the bag to deliver air. The ambu bag’s self-inflating design allows it to snap back in place. Typically you would deliver 10 to 14 “squeezes” per minute for the patient. This works great if you’re in the back of ambulance on the way to a hospital. But for sustained periods, there needs to be a better way.
We saw a clear advantage to automating the hand-held procedure. It would ensure patients continue to get air for days or weeks, and it delivers air more similar to a normal breathing cycle by providing complete control over the motion profile: stroke up, stroke down, controlled velocity, pauses and then repeat. And we kept it cost-efficient.
An electric actuator can recreate the natural tidal volume of air—essentially, what a patient gets in one breath. This is under your complete control with a servo or stepper motor drive. Normal breathing is 5 ml/kg tidal volume (VT), although that varies depending on age, size of the patient and other issues.
Automation with linear actuators
Tolomatic developed two prototypes: one uses an electric rod actuator (ERD15); the other uses a rodless electric actuator (BCS). Both products are screw-driven linear actuators that convert rotary power from a servo motor into linear motion. Rod type actuators include an element (the rod) which extends and retracts from the main body of the actuator. Rodless actuators do not have a rod, rather the travel (stroke) is contained within the actuator’s overall length. Design engineers could use either version.
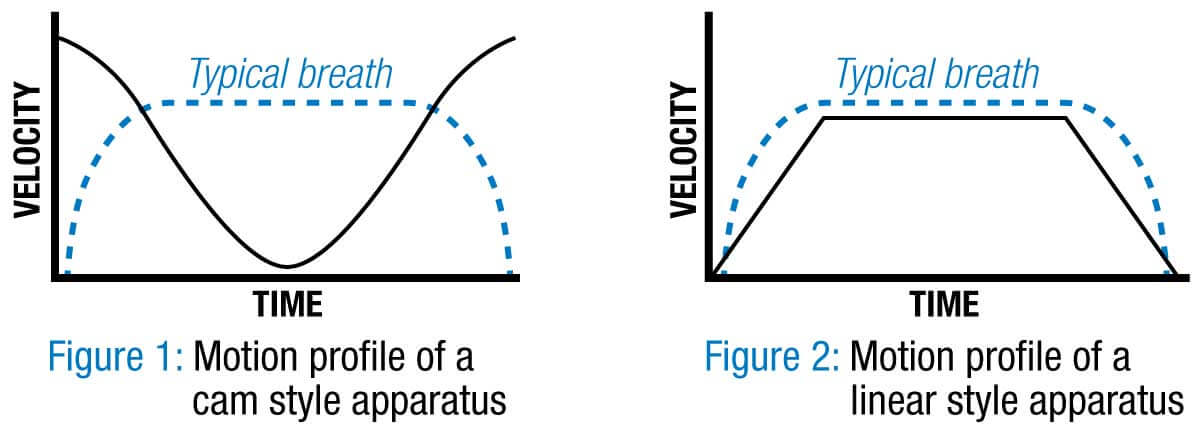
The advantage of screw-driven linear actuators is that they provide smooth and consistent operation–allowing the device to control the velocity, the acceleration and the distance of any move at any point in time. This controlled motion allows for a more continuous volume of air per compression cycle and a more typical breathing cycle.

Many of the open-source designs we saw use an automated single-direction camming solution or pneumatic actuator, but these designs do not allow for varying the stroke length; only the breath frequency. The motion profile for the cam driven solution is counter-intuitive to the natural breathing cycle. The tidal volume of air flow to the patient is always the same: there is a big push at beginning, diminishing airflow at the mid-stroke and then a lot of air pushed through at the completion of the compression cycle. That’s not how most of us breathe.
Linear actuators quickly accelerate up to speed and maintain velocity over a commanded distance. This gives more continuous volumes of air per compression sequence and a more typical breathing cycle. Linear actuator systems can also change the frequency of the induced respirations and also the volume, which is not possible with fixed displacement rotary devices. The total control of motion from a linear electric actuator allows for much more flexible air flow that could now be modified for a patient’s age, size or current needs.
The optimal stroke length for both prototypes is 2 to 4 inches, with less than 25 pounds-of-force minimum required. Both the ERD and BCS design are rated for more than 100 pounds of force. Direct-drive (or in-line) motor configuration simplifies the design and is less expensive. Tolomatic could add gearing to a final design to accommodate a wider variety of motors.

This solution offers reliable long life, and has the ability to integrate in alarm features in case of motor faults, or possible sensors around the airflow quantity and quality.
What’s next
Tolomatic’s prototypes have been operating continuously since April 3. They are quiet, they use food-grade grease, inline motor mounts and stroke lengths from 2.5 to 4 inches. Based on our calculations, the average human takes 8.2 million breaths per year. Our target is a design with at least four million strokes. They are built with Tolomatic’s trade mark Endurance Technology and will last a long time.
Tolomatic has the technical capability to actuate ambu bags; we have electric linear actuators combined with servo motor and drive solutions. The best final solution design would vary depending on whether people want to do 10 or 10,000.
We think we have something to bring to the global need for ventilators. However, we are not medical experts. We are currently creating conversations with partners to finalize a variety of solutions, some that are based on this idea and others that incorporate the electric linear actuator into other design concepts. We continue to look for opportunities to add value to the global shortage of assisted breathing equipment. That’s our hope.
Send us your ideas. Contact:

 Ask an Engineer
Ask an Engineer